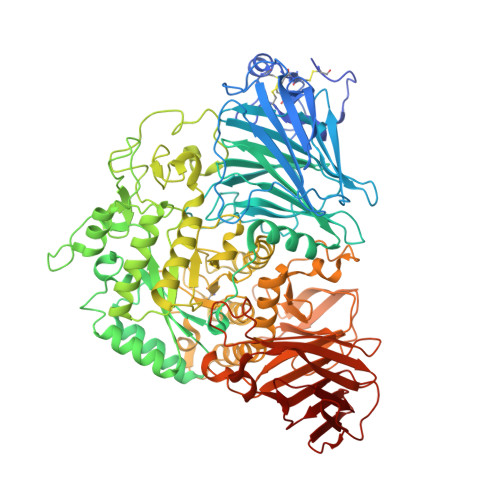
Structural basis for substrate selectivity in human maltase-glucoamylase and sucrase-isomaltase N-terminal domains.
Sim, L., Willemsma, C., Mohan, S., Naim, H.Y., Pinto, B.M., Rose, D.R.(2010) J Biol Chem 285: 17763-17770
- PubMed: 20356844
- DOI: https://doi.org/10.1074/jbc.M109.078980
- Primary Citation of Related Structures:
3LPO, 3LPP - PubMed Abstract:
Human maltase-glucoamylase (MGAM) and sucrase-isomaltase (SI) are small intestinal enzymes that work concurrently to hydrolyze the mixture of linear alpha-1,4- and branched alpha-1,6-oligosaccharide substrates that typically make up terminal starch digestion products. MGAM and SI are each composed of duplicated catalytic domains, N- and C-terminal, which display overlapping substrate specificities. The N-terminal catalytic domain of human MGAM (ntMGAM) has a preference for short linear alpha-1,4-oligosaccharides, whereas N-terminal SI (ntSI) has a broader specificity for both alpha-1,4- and alpha-1,6-oligosaccharides. Here we present the crystal structure of the human ntSI, in apo form to 3.2 A and in complex with the inhibitor kotalanol to 2.15 A resolution. Structural comparison with the previously solved structure of ntMGAM reveals key active site differences in ntSI, including a narrow hydrophobic +1 subsite, which may account for its additional substrate specificity for alpha-1,6 substrates.
Organizational Affiliation:
Department of Medical Biophysics, University of Toronto, Ontario Cancer Institute, Toronto, Ontario M56 2M9, Canada.