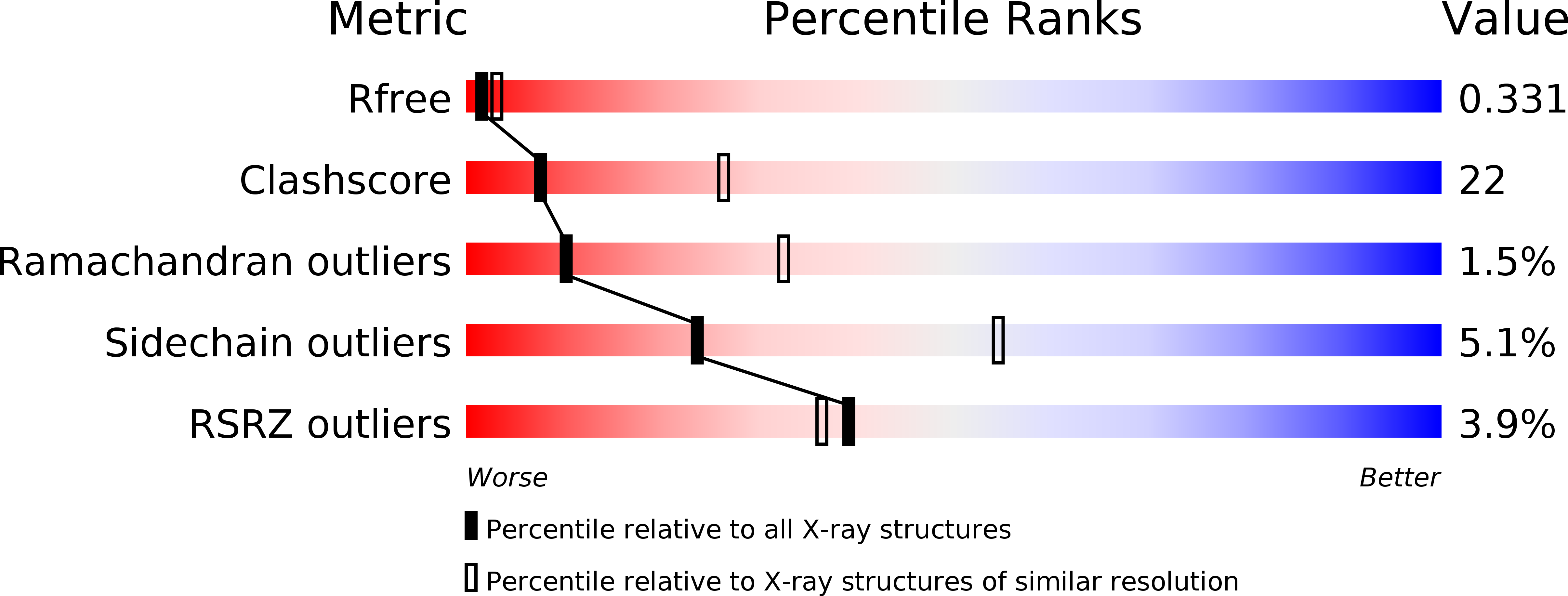
MELK is not necessary for the proliferation of basal-like breast cancer cells.
Huang, H.T., Seo, H.S., Zhang, T., Wang, Y., Jiang, B., Li, Q., Buckley, D.L., Nabet, B., Roberts, J.M., Paulk, J., Dastjerdi, S., Winter, G.E., McLauchlan, H., Moran, J., Bradner, J.E., Eck, M.J., Dhe-Paganon, S., Zhao, J.J., Gray, N.S.(2017) Elife 6
- PubMed: 28926338
- DOI: https://doi.org/10.7554/eLife.26693
- Primary Citation of Related Structures:
5TWL, 5TWU, 5TWY, 5TWZ, 5TX3 - PubMed Abstract:
Thorough preclinical target validation is essential for the success of drug discovery efforts. In this study, we combined chemical and genetic perturbants, including the development of a novel selective maternal embryonic leucine zipper kinase (MELK) inhibitor HTH-01-091, CRISPR/Cas9-mediated MELK knockout, a novel chemical-induced protein degradation strategy, RNA interference and CRISPR interference to validate MELK as a therapeutic target in basal-like breast cancers (BBC). In common culture conditions, we found that small molecule inhibition, genetic deletion, or acute depletion of MELK did not significantly affect cellular growth. This discrepancy to previous findings illuminated selectivity issues of the widely used MELK inhibitor OTSSP167, and potential off-target effects of MELK-targeting short hairpins. The different genetic and chemical tools developed here allow for the identification and validation of any causal roles MELK may play in cancer biology, which will be required to guide future MELK drug discovery efforts. Furthermore, our study provides a general framework for preclinical target validation.
Organizational Affiliation:
Department of Cancer Biology, Dana-Farber Cancer Institute, Boston, United States.