Integrating structure, bioinformatics, and enzymology to discover function: BioH, a new carboxylesterase from Escherichia coli.
Sanishvili, R., Yakunin, A.F., Laskowski, R.A., Skarina, T., Evdokimova, E., Doherty-Kirby, A., Lajoie, G.A., Thornton, J.M., Arrowsmith, C.H., Savchenko, A., Joachimiak, A., Edwards, A.M.(2003) J Biol Chem 278: 26039-26045
- PubMed: 12732651
- DOI: https://doi.org/10.1074/jbc.M303867200
- Primary Citation of Related Structures:
1M33 - PubMed Abstract:
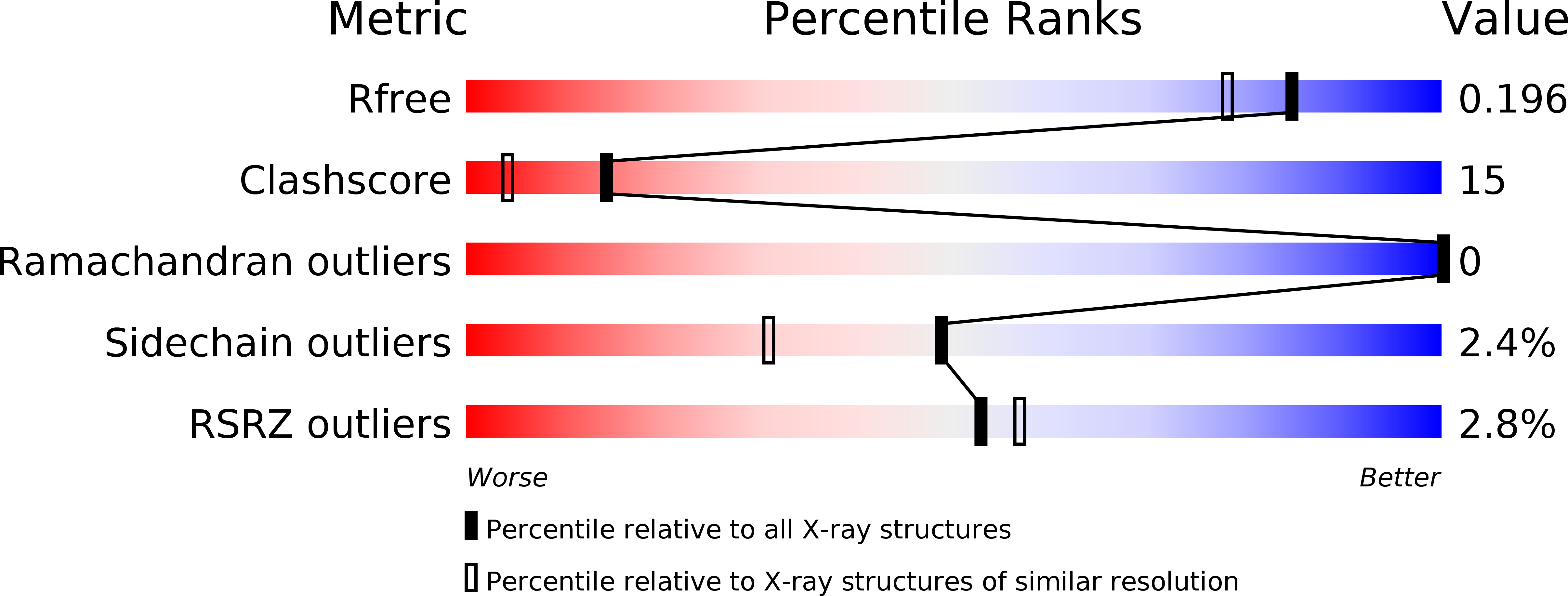
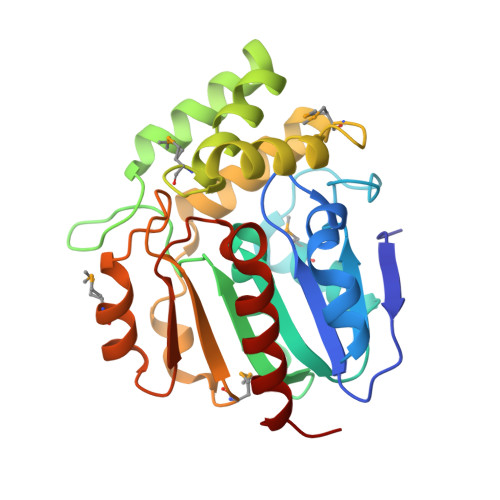
Structural proteomics projects are generating three-dimensional structures of novel, uncharacterized proteins at an increasing rate. However, structure alone is often insufficient to deduce the specific biochemical function of a protein. Here we determined the function for a protein using a strategy that integrates structural and bioinformatics data with parallel experimental screening for enzymatic activity. BioH is involved in biotin biosynthesis in Escherichia coli and had no previously known biochemical function. The crystal structure of BioH was determined at 1.7 A resolution. An automated procedure was used to compare the structure of BioH with structural templates from a variety of different enzyme active sites. This screen identified a catalytic triad (Ser82, His235, and Asp207) with a configuration similar to that of the catalytic triad of hydrolases. Analysis of BioH with a panel of hydrolase assays revealed a carboxylesterase activity with a preference for short acyl chain substrates. The combined use of structural bioinformatics with experimental screens for detecting enzyme activity could greatly enhance the rate at which function is determined from structure.
Organizational Affiliation:
Biosciences Division, Argonne National Laboratory, Argonne, Illinois, 60439, USA.