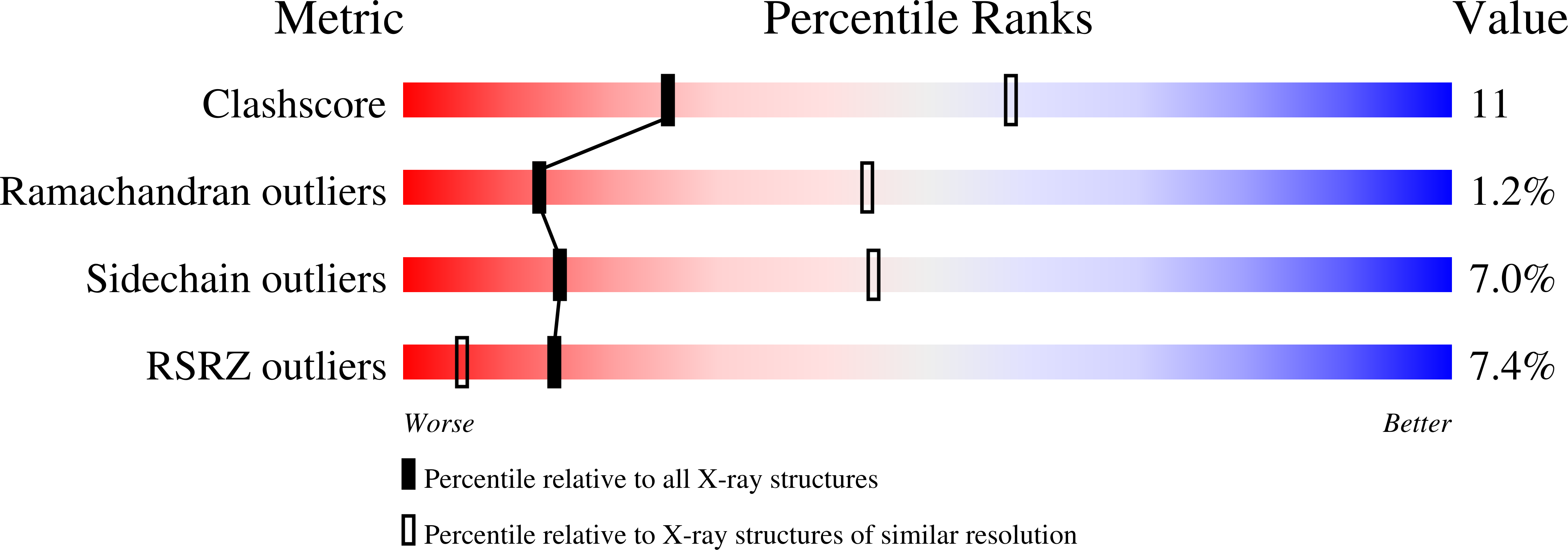
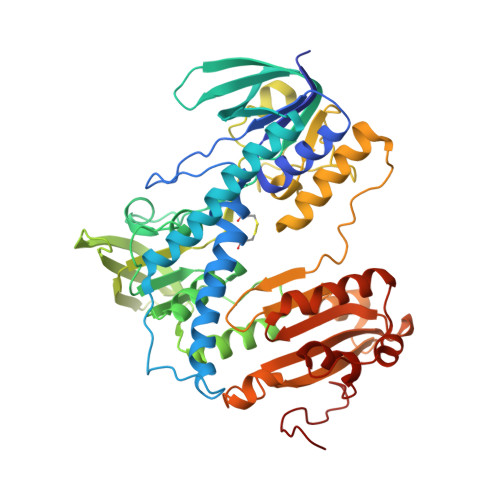
Crystal structure and catalysis of the selenoprotein thioredoxin reductase 1.
Cheng, Q., Sandalova, T., Lindqvist, Y., Arner, E.S.(2009) J Biol Chem 284: 3998-4008
- PubMed: 19054767
- DOI: https://doi.org/10.1074/jbc.M807068200
- Primary Citation of Related Structures:
3EAN, 3EAO - PubMed Abstract:
Selenoproteins contain a highly reactive 21st amino acid selenocysteine (Sec) encoded by recoding of a predefined UGA codon. Because of a lack of selenoprotein supply, high chemical reactivity of Sec, and intricate translation machineries, selenoprotein crystal structures are difficult to obtain. Structural prerequisites for Sec involvement in enzyme catalysis are therefore sparsely known. Here we present the crystal structure of catalytically active rat thioredoxin reductase 1 (TrxR1), revealing surprises at the C-terminal Sec-containing active site in view of previous literature. The oxidized enzyme presents a selenenylsulfide motif in trans-configuration, with the selenium atom of Sec-498 positioned beneath the side chain of Tyr-116, thereby located far from the redox active moieties proposed to be involved in electron transport to the Sec-containing active site. Upon reduction to a selenolthiol motif, the Sec residue moved toward solvent exposure, consistent with its presumed role in reduction of TrxR1 substrates or as target of electrophilic agents inhibiting the enzyme. A Y116I mutation lowered catalytic efficiency in reduction of thioredoxin, but surprisingly increased turnover using 5-hydroxy-1,4-naphthoquinone (juglone) as substrate. The same mutation also decreased sensitivity to inhibition by cisplatin. The results suggest that Tyr-116 plays an important role for catalysis of TrxR1 by interacting with the selenenylsulfide of oxidized TrxR1, thereby facilitating its reduction in the reductive half-reaction of the enzyme. The interaction of a selenenylsulfide with the phenyl ring of a tyrosine, affecting turnover, switch of substrate specificity, and modulation of sensitivity to electrophilic agents, gives important clues into the mechanism of TrxR1, which is a selenoprotein that plays a major role for mammalian cell fate and function. The results also demonstrate that a recombinant selenoprotein TrxR can be produced in high amount and sufficient purity to enable crystal structure determination, which suggests that additional structural studies of these types of proteins are feasible.
Organizational Affiliation:
Division of Biochemistry, Medical Nobel Institute for Biochemistry, Karolinska Institutet, SE-171 77 Stockholm, Sweden.