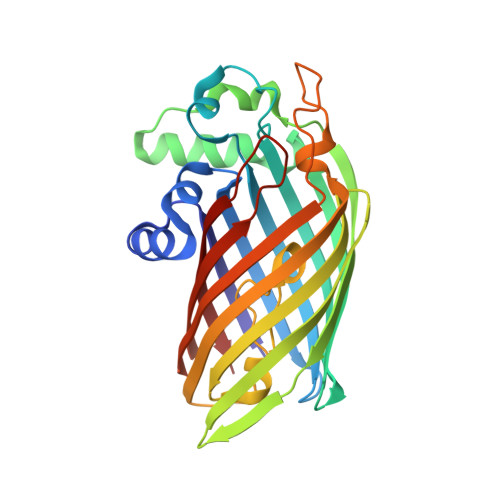
Active-site architecture and catalytic mechanism of the lipid A deacylase LpxR of Salmonella typhimurium
Rutten, L., Mannie, J.-P.B.A., Stead, C.M., Raetz, C.R.H., Reynolds, C.M., Bonvin, A.M.J.J., Tommassen, J.P., Egmond, M.R., Trent, M.S., Gros, P.(2009) Proc Natl Acad Sci U S A 106: 1960-1964
- PubMed: 19174515
- DOI: https://doi.org/10.1073/pnas.0813064106
- Primary Citation of Related Structures:
3FID - PubMed Abstract:
The lipid A portion of lipopolysaccharide, the major component of the outer leaflet of the outer membrane of gram-negative bacteria, is toxic to humans. Modification of lipid A by enzymes often reduces its toxicity. The outer-membrane protein LpxR from Salmonella typhimurium is a lipid A-modifying enzyme. It removes the 3'-acyloxyacyl moiety of the lipid A portion of lipopolysaccharide in a Ca(2+)-dependent manner. Here, we present the crystal structure of S. typhimurium LpxR, crystallized in the presence of zinc ions. The structure, a 12-stranded beta-barrel, reveals that the active site is located between the barrel wall and an alpha-helix formed by an extracellular loop. Based on site-directed mutagenesis and modeling of a substrate on the active site, we propose a catalytic mechanism similar to that of phospholipase A2, in which a Ca(2+) forms the oxyanion hole and a histidine activates a water molecule (or a cascade of two water molecules) that subsequently attacks the carbonyl oxygen of the scissile bond.
Organizational Affiliation:
Laboratory of Crystal and Structural Chemistry, Bijvoet Center for Biomolecular Research, Utrecht University, Utrecht, The Netherlands.