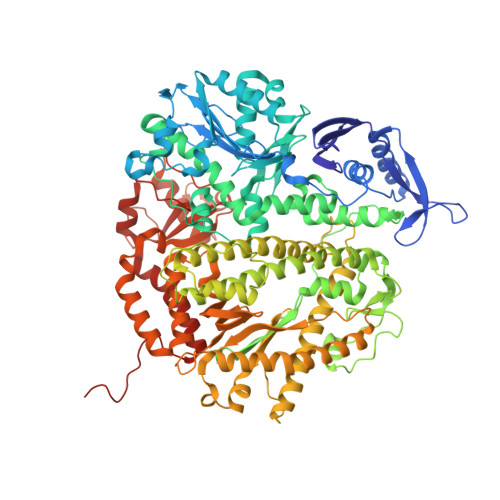
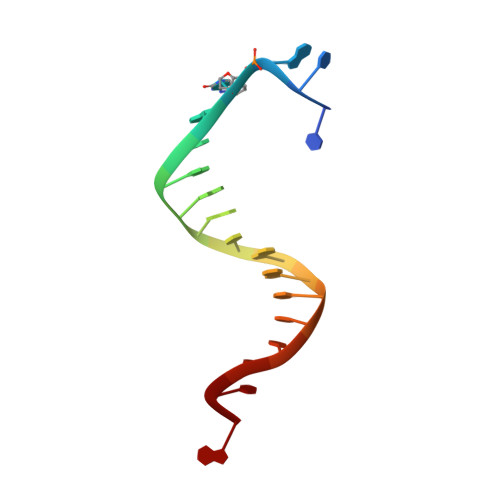
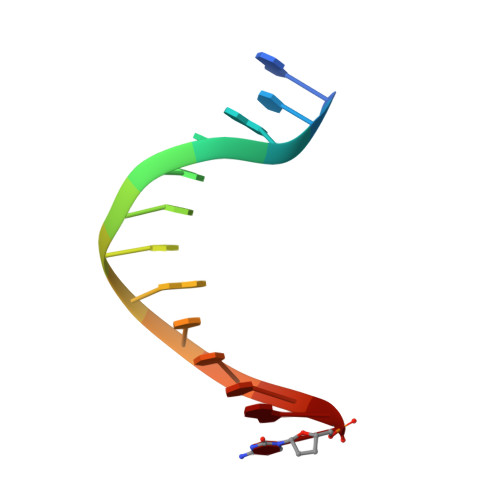
Using a Fluorescent Cytosine Analogue tC(o) To Probe the Effect of the Y567 to Ala Substitution on the Preinsertion Steps of dNMP Incorporation by RB69 DNA Polymerase.
Xia, S., Beckman, J., Wang, J., Konigsberg, W.H.(2012) Biochemistry 51: 4609-4617
- PubMed: 22616982
- DOI: https://doi.org/10.1021/bi300241m
- Primary Citation of Related Structures:
3QNN, 3QNO - PubMed Abstract:
Residues in the nascent base pair binding pocket (NBP) of bacteriophage RB69 DNA polymerase (RB69pol) are responsible for base discrimination. Replacing Tyr567 with Ala leads to greater flexibility in the NBP, increasing the probability of misincorporation. We used the fluorescent cytosine analogue, 1,3-diaza-2-oxophenoxazine (tC(o)), to identify preinsertion step(s) altered by NBP flexibility. When tC(o) is the templating base in a wild-type (wt) RB69pol ternary complex, its fluorescence is quenched only in the presence of dGTP. However, with the RB69pol Y567A mutant, the fluorescence of tC(o) is also quenched in the presence of dATP. We determined the crystal structure of the dATP/tC(o)-containing ternary complex of the RB69pol Y567A mutant at 1.9 Å resolution and found that the incoming dATP formed two hydrogen bonds with an imino-tautomerized form of tC(o). Stabilization of the dATP/tC(o) base pair involved movement of the tC(o) backbone sugar into the DNA minor groove and required tilting of the tC(o) tricyclic ring to prevent a steric clash with L561. This structure, together with the pre-steady-state kinetic parameters and dNTP binding affinity, estimated from equilibrium fluorescence titrations, suggested that the flexibility of the NBP, provided by the Y567 to Ala substitution, led to a more favorable forward isomerization step resulting in an increase in dNTP binding affinity.
Organizational Affiliation:
Department of Molecular Biophysics and Biochemistry, Yale University, New Haven, Connecticut 06520-8114, United States.