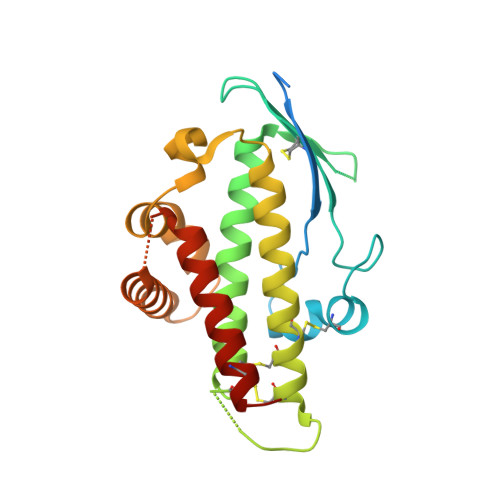
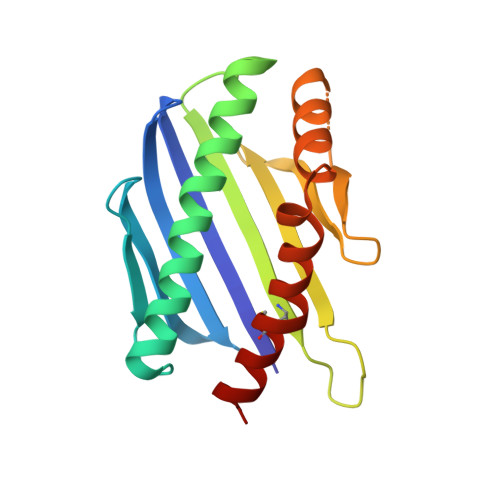
Structural Conservation Despite Huge Sequence Diversity Allows Epcr Binding by the Pfemp1 Family Implicated in Severe Childhood Malaria.
Lau, C.K.Y., Turner, L., Jespersen, J.S., Lowe, E.D., Petersen, B., Wang, C.W., Petersen, J.E.V., Lusingu, J., Theander, T.G., Lavstsen, T., Higgins, M.K.(2015) Cell Host Microbe 17: 118
- PubMed: 25482433
- DOI: https://doi.org/10.1016/j.chom.2014.11.007
- Primary Citation of Related Structures:
4V3D, 4V3E - PubMed Abstract:
The PfEMP1 family of surface proteins is central for Plasmodium falciparum virulence and must retain the ability to bind to host receptors while also diversifying to aid immune evasion. The interaction between CIDRα1 domains of PfEMP1 and endothelial protein C receptor (EPCR) is associated with severe childhood malaria. We combine crystal structures of CIDRα1:EPCR complexes with analysis of 885 CIDRα1 sequences, showing that the EPCR-binding surfaces of CIDRα1 domains are conserved in shape and bonding potential, despite dramatic sequence diversity. Additionally, these domains mimic features of the natural EPCR ligand and can block this ligand interaction. Using peptides corresponding to the EPCR-binding region, antibodies can be purified from individuals in malaria-endemic regions that block EPCR binding of diverse CIDRα1 variants. This highlights the extent to which such a surface protein family can diversify while maintaining ligand-binding capacity and identifies features that should be mimicked in immunogens to prevent EPCR binding.
Organizational Affiliation:
Department of Biochemistry, University of Oxford, South Parks Road, OX1 3QU Oxford, UK.