X-ray structure of Phosphoglycerate Mutase 1(PGAM1) complexed with a small molecule
Zhou, L., Jiang, L.L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Phosphoglycerate mutase 1 | A [auth B], B [auth C] | 262 | Homo sapiens | Mutation(s): 0 Gene Names: PGAM1, PGAMA, CDABP0006 EC: 5.4.2.11 (PDB Primary Data), 5.4.2.4 (PDB Primary Data) | 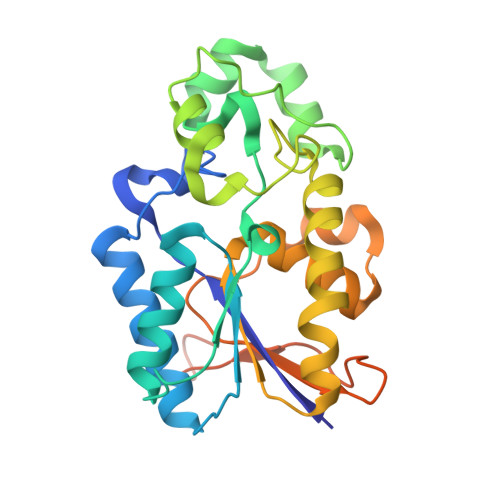 |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P18669 (Homo sapiens) Explore P18669 Go to UniProtKB: P18669 | |||||
PHAROS: P18669 GTEx: ENSG00000171314 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P18669 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AZN Query on AZN | D [auth B], E [auth C] | ALIZARIN RED C14 H8 O7 S JKYKXTRKURYNGW-UHFFFAOYSA-N |  | ||
| MES Query on MES | F [auth C] | 2-(N-MORPHOLINO)-ETHANESULFONIC ACID C6 H13 N O4 S SXGZJKUKBWWHRA-UHFFFAOYSA-N |  | ||
| CL Query on CL | C [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 82.64 | α = 90 |
| b = 85.267 | β = 90 |
| c = 102.466 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SCALEPACK | data scaling |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| PHASER | phasing |