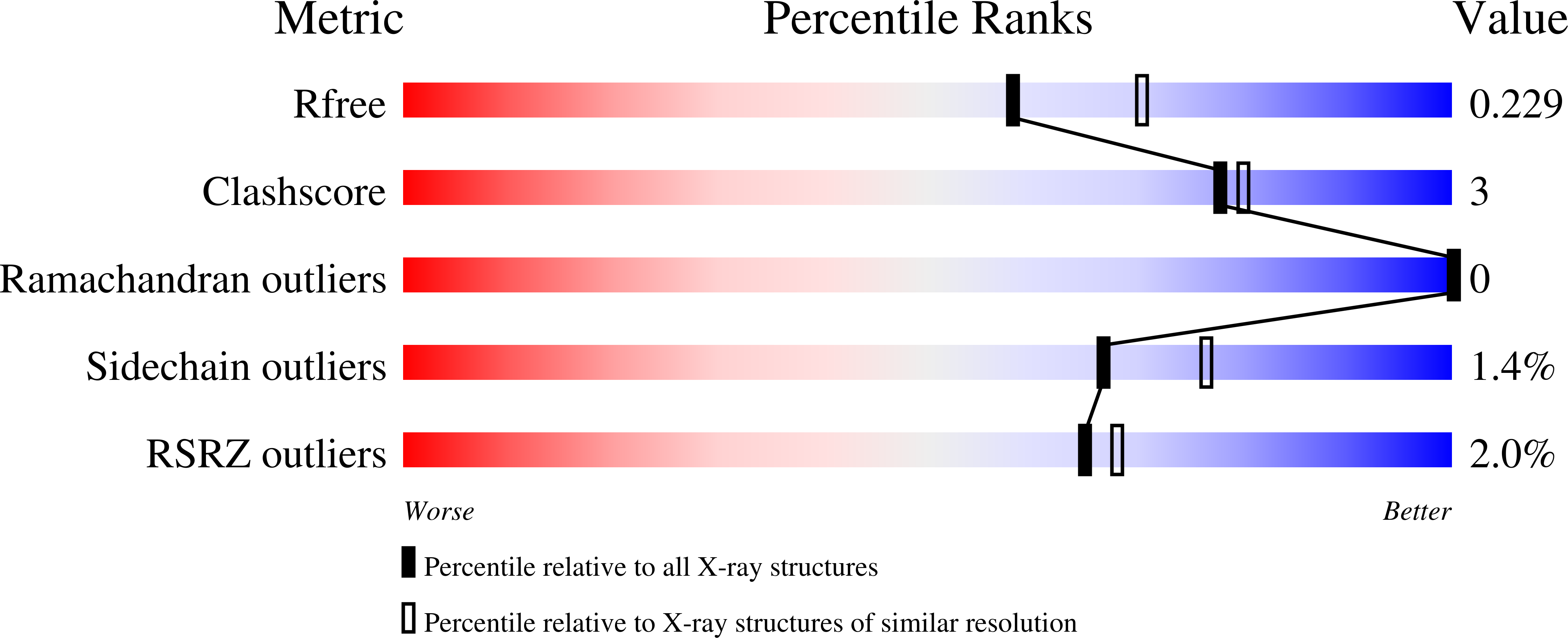
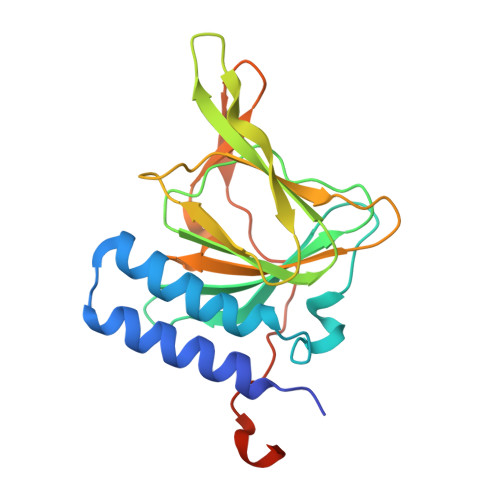
Structure of 3-mercaptopropionic acid dioxygenase with a substrate analog reveals bidentate substrate binding at the iron center.
York, N.J., Lockart, M.M., Sardar, S., Khadka, N., Shi, W., Stenkamp, R.E., Zhang, J., Kiser, P.D., Pierce, B.S.(2021) J Biol Chem 296: 100492-100492
- PubMed: 33662397
- DOI: https://doi.org/10.1016/j.jbc.2021.100492
- Primary Citation of Related Structures:
6XB9, 7KOV - PubMed Abstract:
Thiol dioxygenases are a subset of nonheme iron oxygenases that catalyze the formation of sulfinic acids from sulfhydryl-containing substrates and dioxygen. Among this class, cysteine dioxygenases (CDOs) and 3-mercaptopropionic acid dioxygenases (3MDOs) are the best characterized, and the mode of substrate binding for CDOs is well understood. However, the manner in which 3-mercaptopropionic acid (3MPA) coordinates to the nonheme iron site in 3MDO remains a matter of debate. A model for bidentate 3MPA coordination at the 3MDO Fe-site has been proposed on the basis of computational docking, whereas steady-state kinetics and EPR spectroscopic measurements suggest a thiolate-only coordination of the substrate. To address this gap in knowledge, we determined the structure of Azobacter vinelandii 3MDO (Av3MDO) in complex with the substrate analog and competitive inhibitor, 3-hydroxypropionic acid (3HPA). The structure together with DFT computational modeling demonstrates that 3HPA and 3MPA associate with iron as chelate complexes with the substrate-carboxylate group forming an additional interaction with Arg168 and the thiol bound at the same position as in CDO. A chloride ligand was bound to iron in the coordination site assigned as the O 2 -binding site. Supporting HYSCORE spectroscopic experiments were performed on the (3MPA/NO)-bound Av3MDO iron nitrosyl (S = 3/2) site. In combination with spectroscopic simulations and optimized DFT models, this work provides an experimentally verified model of the Av3MDO enzyme-substrate complex, effectively resolving a debate in the literature regarding the preferred substrate-binding denticity. These results elegantly explain the observed 3MDO substrate specificity, but leave unanswered questions regarding the mechanism of substrate-gated reactivity with dioxygen.
Organizational Affiliation:
Department of Chemistry & Biochemistry, University of Alabama, Tuscaloosa, Alabama, USA.