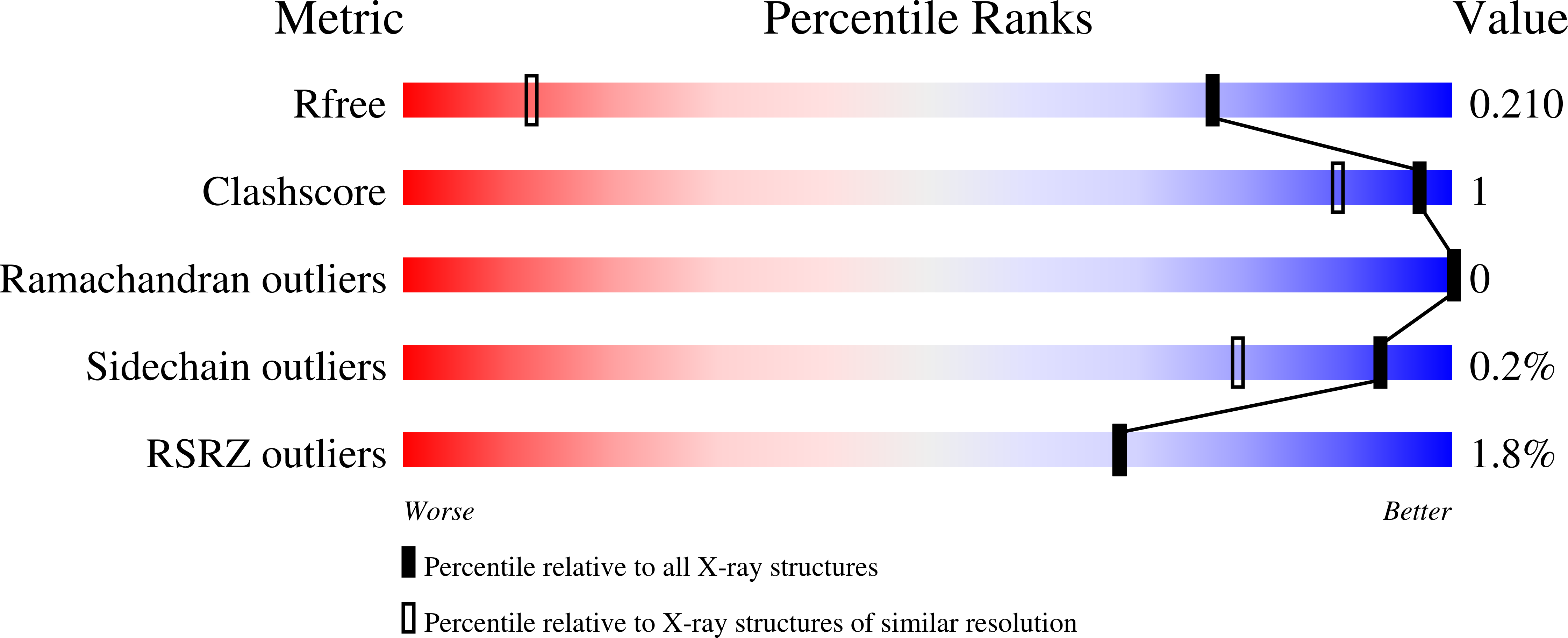
Alternative binding modes in abortive NADH-alcohol complexes of horse liver alcohol dehydrogenase.
Plapp, B.V., Subramanian, R.(2021) Arch Biochem Biophys 701: 108825-108825
- PubMed: 33675814
- DOI: https://doi.org/10.1016/j.abb.2021.108825
- Primary Citation of Related Structures:
6XT2, 7JQA, 7K35 - PubMed Abstract:
Enzymes typically have high specificity for their substrates, but the structures of substrates and products differ, and multiple modes of binding are observed. In this study, high resolution X-ray crystallography of complexes with NADH and alcohols show alternative modes of binding in the active site. Enzyme crystallized with the good substrates NAD + and 4-methylbenzyl alcohol was found to be an abortive complex of NADH with 4-methylbenzyl alcohol rotated to a "non-productive" mode as compared to the structures that resemble reactive Michaelis complexes with NAD + and 2,2,2-trifluoroethanol or 2,3,4,5,6-pentafluorobenzyl alcohol. The NADH is formed by reduction of the NAD + with the alcohol during the crystallization. The same structure was also formed by directly crystallizing the enzyme with NADH and 4-methylbenzyl alcohol. Crystals prepared with NAD + and 4-bromobenzyl alcohol also form the abortive complex with NADH. Surprisingly, crystals prepared with NAD + and the strong inhibitor 1H,1H-heptafluorobutanol also had NADH, and the alcohol was bound in two different conformations that illustrate binding flexibility. Oxidation of 2-methyl-2,4-pentanediol during the crystallization apparently led to reduction of the NAD + . Kinetic studies show that high concentrations of alcohols can bind to the enzyme-NADH complex and activate or inhibit the enzyme. Together with previous studies on complexes with NADH and formamide analogues of the carbonyl substrates, models for the Michaelis complexes with NAD + -alcohol and NADH-aldehyde are proposed.
Organizational Affiliation:
Department of Biochemistry, The University of Iowa, Iowa City, IA, 52242, USA. Electronic address: [email protected].