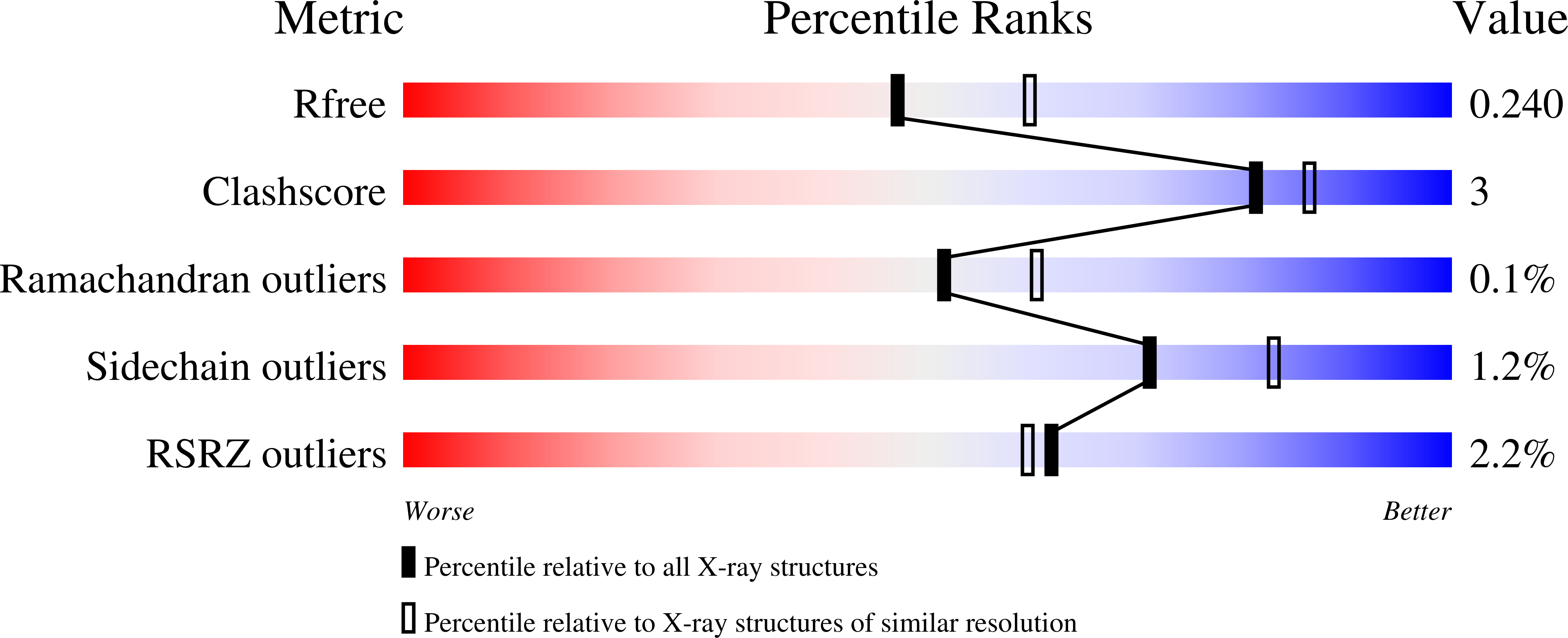
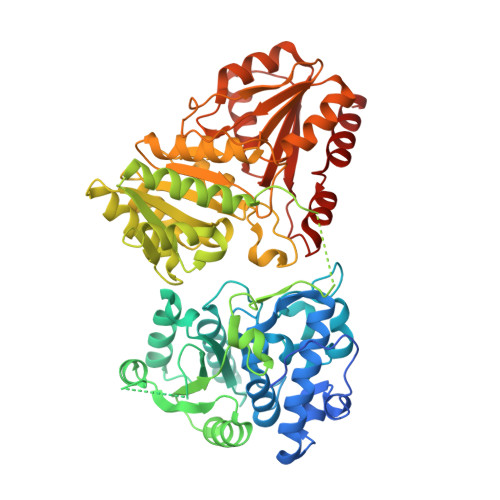
1-deoxy-D-xylulose-5-phosphate synthase from Pseudomonas aeruginosa and Klebsiella pneumoniae reveals conformational changes upon cofactor binding.
Hamid, R., Adam, S., Lacour, A., Monjas, L., Kohnke, J., Hirsch, A.K.H.(2023) J Biol Chem 299: 105152-105152
- PubMed: 37567475
- DOI: https://doi.org/10.1016/j.jbc.2023.105152
- Primary Citation of Related Structures:
8A29, 8A45, 8A4D, 8A5K - PubMed Abstract:
The ESKAPE bacteria are the six highly virulent and antibiotic-resistant pathogens that require the most urgent attention for the development of novel antibiotics. Detailed knowledge of target proteins specific to bacteria is essential to develop novel treatment options. The methylerythritol-phosphate (MEP) pathway, which is absent in humans, represents a potentially valuable target for the development of novel antibiotics. Within the MEP pathway, the enzyme 1-deoxy-D-xylulose-5-phosphate synthase (DXPS) catalyzes a crucial, rate-limiting first step and a branch point in the biosynthesis of the vitamins B1 and B6. We report the high-resolution crystal structures of DXPS from the important ESKAPE pathogens Pseudomonas aeruginosa and Klebsiella pneumoniae in both the co-factor-bound and the apo forms. We demonstrate that the absence of the cofactor thiamine diphosphate results in conformational changes that lead to disordered loops close to the active site that might be important for the design of potent DXPS inhibitors. Collectively, our results provide important structural details that aid in the assessment of DXPS as a potential target in the ongoing efforts to combat antibiotic resistance.
Organizational Affiliation:
Department of Drug Design and Optimization, Helmholtz Institute for Pharmaceutical Research Saarland (HIPS) - Helmholtz Centre for Infection Research (HZI), Saarbrücken, Germany; Department of Pharmacy, Saarland University, Saarbrücken, Germany.