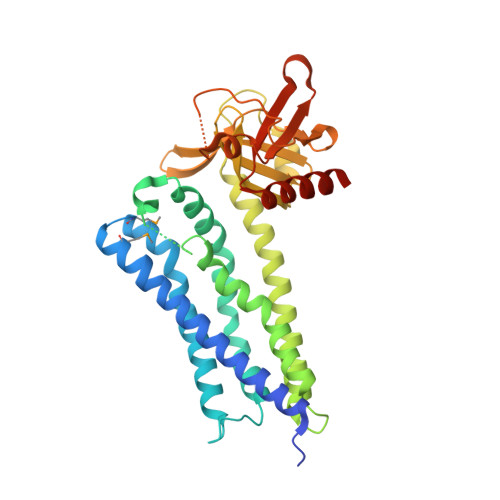
Structure of the Rho-specific guanine nucleotide-exchange factor Xpln
Murayama, K., Kato-Murayama, M., Akasaka, R., Terada, T., Yokoyama, S., Shirouzu, M.(2012) Acta Crystallogr Sect F Struct Biol Cryst Commun 68: 1455-1459
- PubMed: 23192023
- DOI: https://doi.org/10.1107/S1744309112045265
- Primary Citation of Related Structures:
2Z0Q - PubMed Abstract:
Xpln is a guanine nucleotide-exchange factor (GEF) for Rho GTPases. A Dbl homology (DH) domain followed by a pleckstrin homology (PH) domain is a widely adopted GEF-domain architecture. The Xpln structure solely comprises these two domains. Xpln activates RhoA and RhoB, but not RhoC, although their GTPase sequences are highly conserved. The molecular mechanism of the selectivity of Xpln for Rho GTPases is still unclear. In this study, the crystal structure of the tandemly arranged DH-PH domains of mouse Xpln, with a single molecule in the asymmetric unit, was determined at 1.79 Å resolution by the multiwavelength anomalous dispersion method. The DH-PH domains of Xpln share high structural similarity with those from neuroepithelial cell-transforming gene 1 protein, PDZ-RhoGEF, leukaemia-associated RhoGEF and intersectins 1 and 2. The crystal structure indicated that the α4-α5 loop in the DH domain is flexible and that the DH and PH domains interact with each other intramolecularly, thus suggesting that PH-domain rearrangement occurs upon RhoA binding.
Organizational Affiliation:
Graduate School of Biomedical Engineering, Tohoku University, 2-1 Seiryo, Aoba, Sendai 980-8575, Japan.