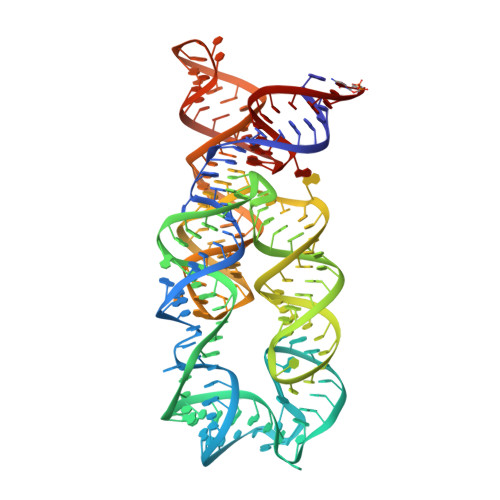
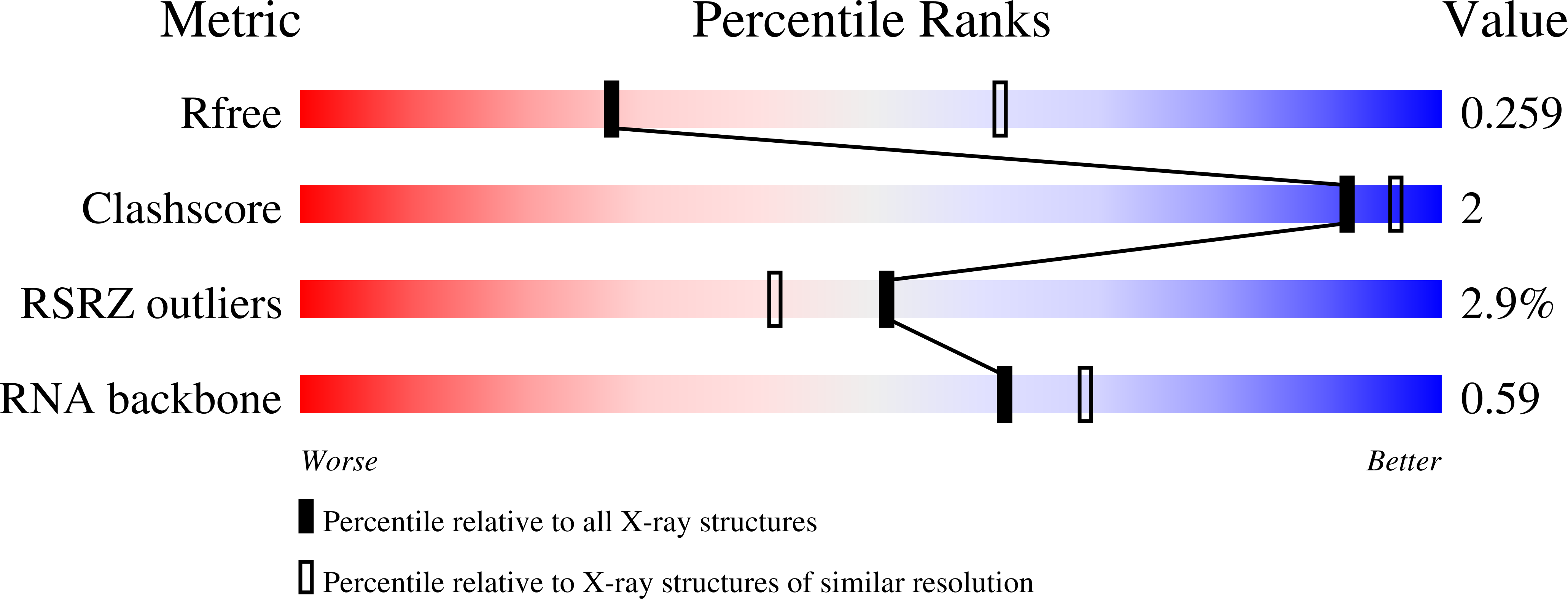Structural insights into amino acid binding and gene control by a lysine riboswitch.
Serganov, A., Huang, L., Patel, D.J.(2008) Nature 455: 1263-1267
- PubMed: 18784651
- DOI: https://doi.org/10.1038/nature07326
- Primary Citation of Related Structures:
3DIG, 3DIL, 3DIM, 3DIO, 3DIQ, 3DIR, 3DIS, 3DIX, 3DIY, 3DIZ, 3DJ0, 3DJ2 - PubMed Abstract:
In bacteria, the intracellular concentration of several amino acids is controlled by riboswitches. One of the important regulatory circuits involves lysine-specific riboswitches, which direct the biosynthesis and transport of lysine and precursors common for lysine and other amino acids. To understand the molecular basis of amino acid recognition by riboswitches, here we present the crystal structure of the 174-nucleotide sensing domain of the Thermotoga maritima lysine riboswitch in the lysine-bound (1.9 ångström (A)) and free (3.1 A) states. The riboswitch features an unusual and intricate architecture, involving three-helical and two-helical bundles connected by a compact five-helical junction and stabilized by various long-range tertiary interactions. Lysine interacts with the junctional core of the riboswitch and is specifically recognized through shape-complementarity within the elongated binding pocket and through several direct and K(+)-mediated hydrogen bonds to its charged ends. Our structural and biochemical studies indicate preformation of the riboswitch scaffold and identify conformational changes associated with the formation of a stable lysine-bound state, which prevents alternative folding of the riboswitch and facilitates formation of downstream regulatory elements. We have also determined several structures of the riboswitch bound to different lysine analogues, including antibiotics, in an effort to understand the ligand-binding capabilities of the lysine riboswitch and understand the nature of antibiotic resistance. Our results provide insights into a mechanism of lysine-riboswitch-dependent gene control at the molecular level, thereby contributing to continuing efforts at exploration of the pharmaceutical and biotechnological potential of riboswitches.
Organizational Affiliation:
Structural Biology Program, Memorial Sloan-Kettering Cancer Center, New York, New York 10065, USA. [email protected]