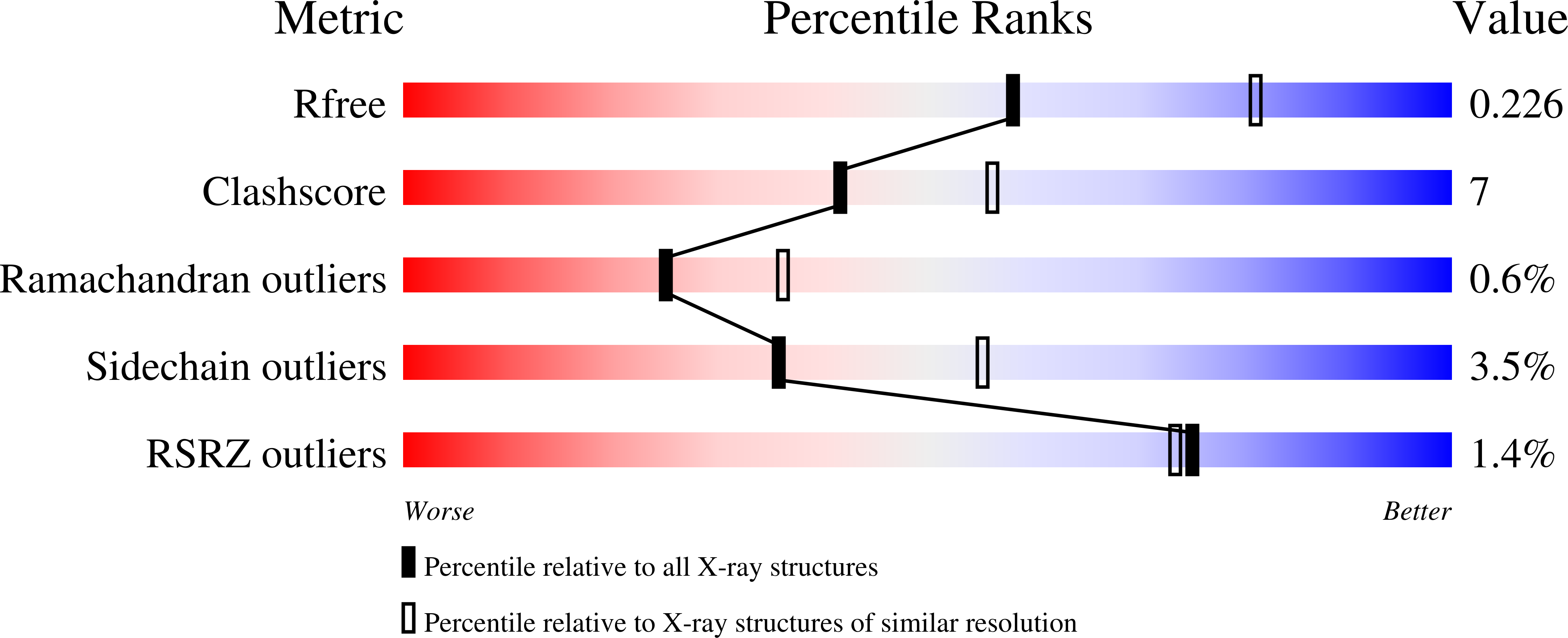
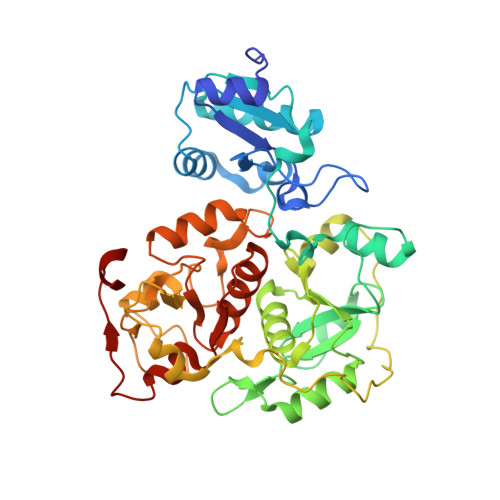
Crystal structure of YnjE from Escherichia coli, a sulfurtransferase with three rhodanese domains.
Hanzelmann, P., Dahl, J.U., Kuper, J., Urban, A., Muller-Theissen, U., Leimkuhler, S., Schindelin, H.(2009) Protein Sci 18: 2480-2491
- PubMed: 19798741
- DOI: https://doi.org/10.1002/pro.260
- Primary Citation of Related Structures:
3IPO, 3IPP - PubMed Abstract:
Rhodaneses/sulfurtransferases are ubiquitous enzymes that catalyze the transfer of sulfane sulfur from a donor molecule to a thiophilic acceptor via an active site cysteine that is modified to a persulfide during the reaction. Here, we present the first crystal structure of a triple-domain rhodanese-like protein, namely YnjE from Escherichia coli, in two states where its active site cysteine is either unmodified or present as a persulfide. Compared to well-characterized tandem domain rhodaneses, which are composed of one inactive and one active domain, YnjE contains an extra N-terminal inactive rhodanese-like domain. Phylogenetic analysis reveals that YnjE triple-domain homologs can be found in a variety of other gamma-proteobacteria, in addition, some single-, tandem-, four and even six-domain variants exist. All YnjE rhodaneses are characterized by a highly conserved active site loop (CGTGWR) and evolved independently from other rhodaneses, thus forming their own subfamily. On the basis of structural comparisons with other rhodaneses and kinetic studies, YnjE, which is more similar to thiosulfate:cyanide sulfurtransferases than to 3-mercaptopyruvate:cyanide sulfurtransferases, has a different substrate specificity that depends not only on the composition of the active site loop with the catalytic cysteine at the first position but also on the surrounding residues. In vitro YnjE can be efficiently persulfurated by the cysteine desulfurase IscS. The catalytic site is located within an elongated cleft, formed by the central and C-terminal domain and is lined by bulky hydrophobic residues with the catalytic active cysteine largely shielded from the solvent.
Organizational Affiliation:
Rudolf Virchow Center for Experimental Biomedicine, University of Würzburg, 97080 Würzburg, Germany. [email protected]