Comparative structural and functional analysis of phi class glutathione transferases involved in multiple-herbicide resistance of grass weeds and crops.
Georgakis, N., Poudel, N., Papageorgiou, A.C., Labrou, N.E.(2020) Plant Physiol Biochem 149: 266-276
- PubMed: 32088578
- DOI: https://doi.org/10.1016/j.plaphy.2020.02.012
- Primary Citation of Related Structures:
6RIV - PubMed Abstract:
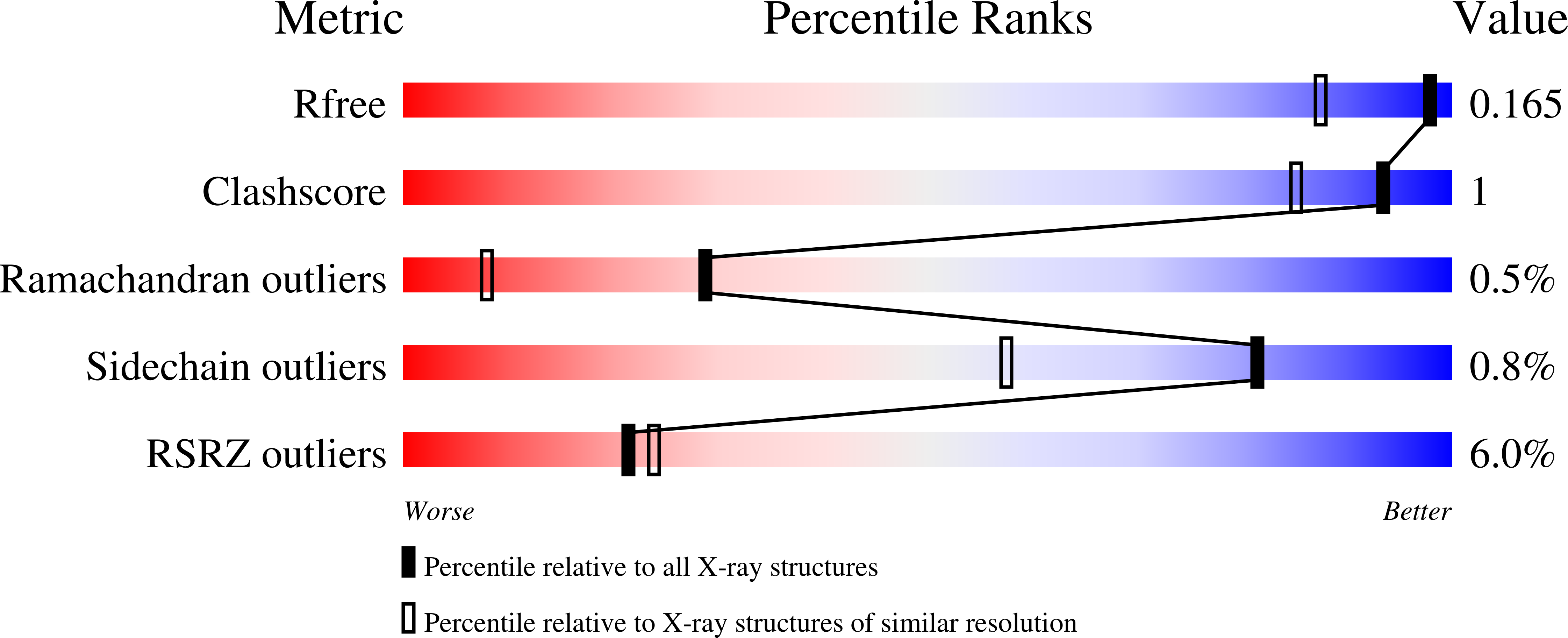
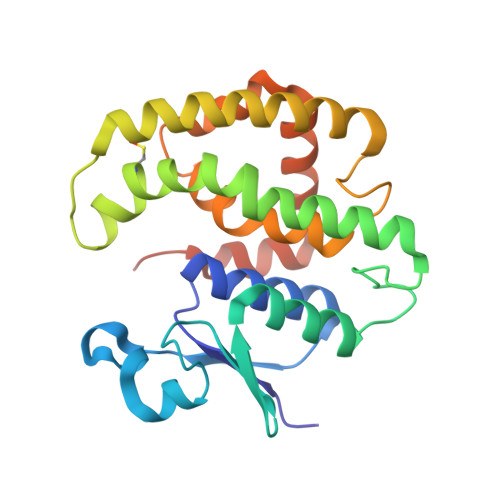
Multiple-herbicide resistant (MHR) weeds are a global problem and a looming threat to weed control in crops. MHR weeds express a specific phi class glutathione transferase (MHR-GSTF) which seems to contribute to herbicide resistance. The present work aims to investigate the structure and catalytic properties of the MHR-GSTFs from different grass weeds and crops (Alopecurus myosuroides, Lolium rigidum, Hordeum vulgare, Triticum aestivum). Recombinant MHR-GSTFs were expressed in E. coli and purified by affinity chromatography. Kinetic analysis of substrate specificity using a range of thiol substrates and xenobiotic compounds suggested that all enzymes display a broad range of specificity and are capable of detoxifying major stress-induced toxic products. Notably, all tested enzymes exhibited high activity towards organic hydroperoxides. The crystal structure of MHR-GSTF from Alopecurus myosuroides (AmGSTF) was determined by molecular replacement at 1.33 Å resolution. The enzyme was resolved with bound glutathione sulfenic acid (GSOH) at the G-site and succinic acid at the H-site. The enzyme shows conserved structural features compared to other Phi class GSTs. However, some differences were observed at the C-terminal helix H9 that may affect substrate specificity. The structural and functional features of AmGSTF were compared with those of the homologue crop enzymes (HvGSTF and TaGSTF) and discussed in light of their contribution to the MHR mechanism.
Organizational Affiliation:
Laboratory of Enzyme Technology, Department of Biotechnology, School of Applied Biology and Biotechnology, Agricultural University of Athens, 75 Iera Odos Street, GR-11855, Athens, Greece.