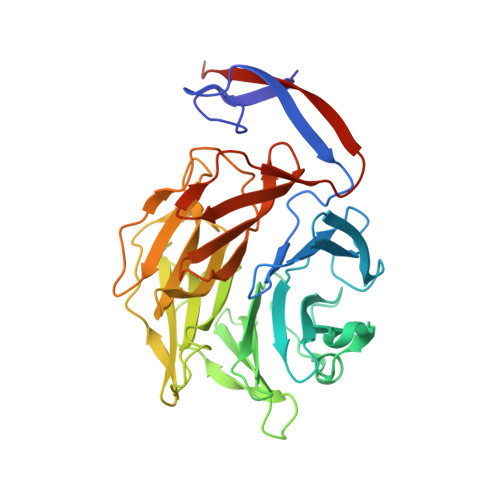
Structural insights into the fungi-nematodes interaction mediated by fucose-specific lectin AofleA from Arthrobotrys oligospora.
Liu, M., Cheng, X., Wang, J., Tian, D., Tang, K., Xu, T., Zhang, M., Wang, Y., Wang, M.(2020) Int J Biol Macromol 164: 783-793
- PubMed: 32698064
- DOI: https://doi.org/10.1016/j.ijbiomac.2020.07.173
- Primary Citation of Related Structures:
7C37, 7C38, 7C39, 7C3C, 7C3D, 7C3E - PubMed Abstract:
Fungal lectin can bind specific carbohydrate structures of the host and work in recognition and adhesion or as a toxic factor. AofleA, as a fucose-specific lectin from widely studied nematode predatory fungus Arthrobotrys oligospora, possibly plays a key role in the event of capturing nematodes, but the mechanism remains unknown. Here we report the crystal structure of AofleA, which exists as a homodimer with each subunit folds as a six-bladed β-propeller. Our structural and biological results revealed that three of the six putative binding sites of AofleA had fucose-binding abilities. In addition, we found that AofleA could bind to the pharynx and intestine of the nematode in a fucose-binding-dependent manner. Our results facilitate the understanding of the mechanism that fucose-specific lectin mediates fungi-nematodes interaction, and provide structural information for the development of potential applications of AofleA.
Organizational Affiliation:
School of Life Sciences, Anhui University, Hefei 230601, Anhui, China; Institutes of Physical Science and Information Technology, Anhui University, Hefei 230601, Anhui, China; Key Laboratory of Human Microenvironment and Precision Medicine of Anhui Higher Education Institutes, Anhui University, Hefei 230601, Anhui, China.