Mobocertinib (TAK-788): A Targeted Inhibitor of EGFR Exon 20 Insertion Mutants in Non-Small Cell Lung Cancer.
Gonzalvez, F., Vincent, S., Baker, T.E., Gould, A.E., Li, S., Wardwell, S.D., Nadworny, S., Ning, Y., Zhang, S., Huang, W.S., Hu, Y., Li, F., Greenfield, M.T., Zech, S.G., Das, B., Narasimhan, N.I., Clackson, T., Dalgarno, D., Shakespeare, W.C., Fitzgerald, M., Chouitar, J., Griffin, R.J., Liu, S., Wong, K.K., Zhu, X., Rivera, V.M.(2021) Cancer Discov 11: 1672-1687
- PubMed: 33632773
- DOI: https://doi.org/10.1158/2159-8290.CD-20-1683
- Primary Citation of Related Structures:
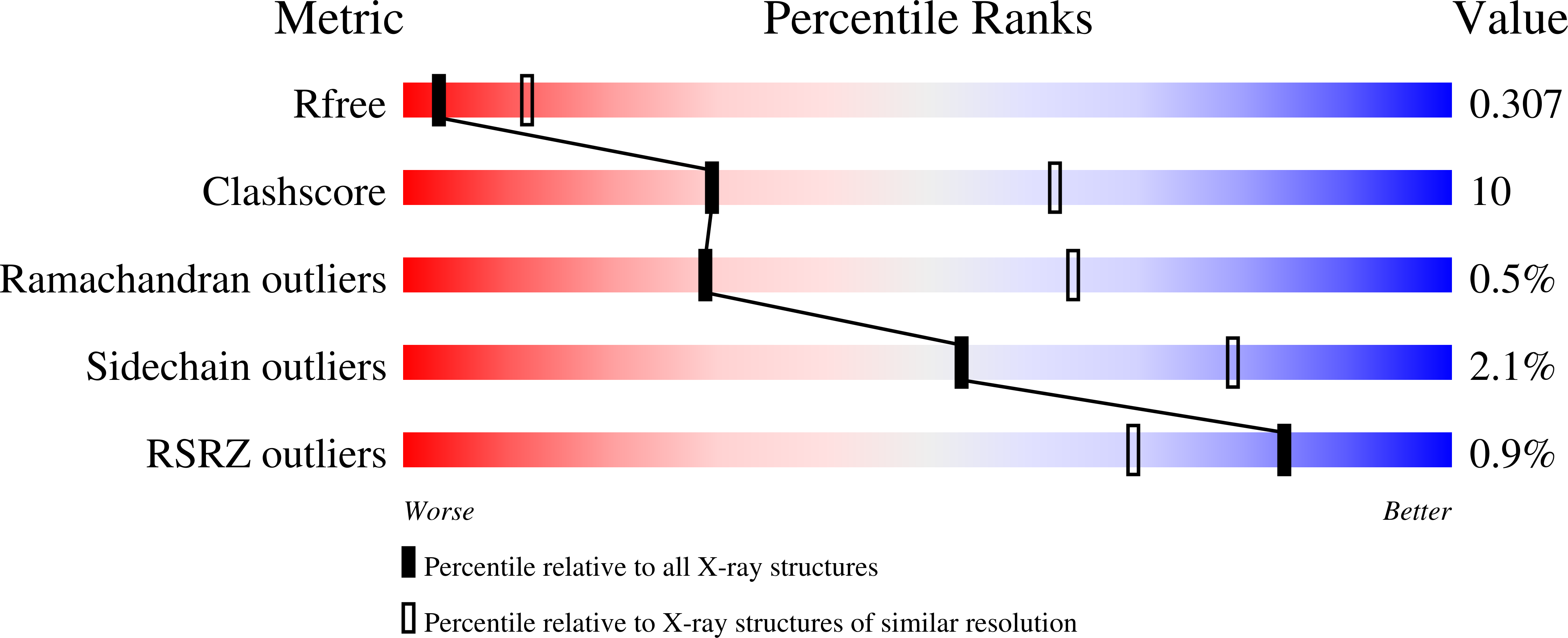
7LGS - PubMed Abstract:
Most EGFR exon 20 insertion ( EGFR ex20ins) driver mutations in non-small cell lung cancer (NSCLC) are insensitive to approved EGFR tyrosine kinase inhibitors (TKI). To address the limitations of existing therapies targeting EGFR -mutated NSCLC, mobocertinib (TAK-788), a novel irreversible EGFR TKI, was specifically designed to potently inhibit oncogenic variants containing activating EGFR ex20ins mutations with selectivity over wild-type EGFR. The in vitro and in vivo activity of mobocertinib was evaluated in engineered and patient-derived models harboring diverse EGFR ex20ins mutations. Mobocertinib inhibited viability of various EGFRex20ins-driven cell lines more potently than approved EGFR TKIs and demonstrated in vivo antitumor efficacy in patient-derived xenografts and murine orthotopic models. These findings support the ongoing clinical development of mobocertinib for the treatment of EGFR ex20ins-mutated NSCLC. SIGNIFICANCE: No oral EGFR-targeted therapies are approved for EGFR exon 20 insertion ( EGFR ex20ins) mutation-driven NSCLC. Mobocertinib is a novel small-molecule EGFR inhibitor specifically designed to target EGFRex20ins mutants. Preclinical data reported here support the clinical development of mobocertinib in patients with NSCLC with EGFR exon 20 insertion mutations. See related commentary by Pacheco, p. 1617 . This article is highlighted in the In This Issue feature, p. 1601 .
Organizational Affiliation:
ARIAD Pharmaceuticals, Inc., Cambridge, Massachusetts, a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.