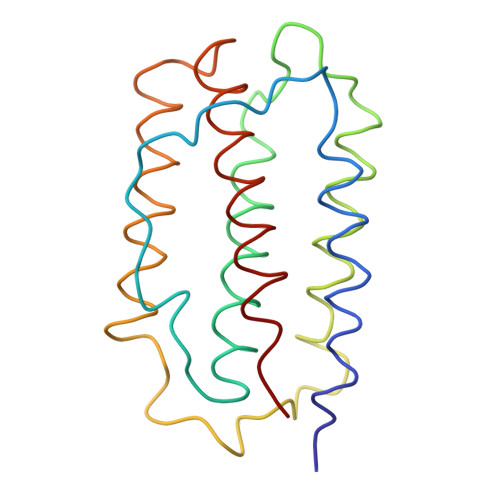
Three-dimensional crystal structure of recombinant murine interferon-beta.
Senda, T., Shimazu, T., Matsuda, S., Kawano, G., Shimizu, H., Nakamura, K.T., Mitsui, Y.(1992) EMBO J 11: 3193-3201
- PubMed: 1505514
- DOI: https://doi.org/10.1002/j.1460-2075.1992.tb05396.x
- Primary Citation of Related Structures:
1IFA - PubMed Abstract:
The crystal structure of recombinant murine interferon-beta (IFN-beta) has been solved by the multiple isomorphous replacement method and refined to an R-factor of 20.5% against 2.6 A X-ray diffraction data. The structure shows a variant of the alpha-helix bundle with a new chain-folding topology, which seems to represent a basic structural framework of all the IFN-alpha and IFN-beta molecules belonging to the type I family. Functionally important segments of the polypeptide chain, as implied through numerous gene manipulation studies carried out so far, are spatially clustered indicating the binding site(s) to the receptor(s). Comparison of the present structure with those of other alpha-helical cytokine proteins, including porcine growth hormone, interleukin 2 and interferon gamma, indicated either a topological similarity in chain folding or a similar spatial arrangement of the alpha-helices.
Organizational Affiliation:
Faculty of Engineering, Nagaoka University of Technology, Niigata, Japan.